Overview
A digital health coaching company partnered with a major pharmaceutical client to address a critical gap: patients starting GLP-1 medication therapy were dropping out of a structured twelve-week coaching program before experiencing its benefits. Roughly half of enrolled patients left within the first week, and attrition continued steeply through weeks two and three.
This case study describes how I identified the root causes of early attrition, designed a regulatory-compliant intervention that had never been attempted in this format before, and navigated a sixteen-month approval process as the point person for content design, evidence research, MLR review, and client communications to launch branded educational modules within a white label coaching program.
Client and Context
A major pharmaceutical company had launched a white label digital coaching program to support patients managing type 2 diabetes with a GLP-1 medication. The twelve-week program used a hybrid coaching model: weekly phone calls with a dedicated health coach, plus a digital experience of educational articles, videos, and patient-reported outcomes delivered through a web and mobile app. Educational content was hosted across two platforms, with the coaching company producing original materials and the pharmaceutical client providing branded patient resources through its own consumer-facing website.
The program was unbranded by design, giving the client flexibility to deploy it across multiple contexts. However, this created a significant gap. Because the coaching content couldn’t directly address medication-specific questions, patients were left without answers to the things they cared about most in those first critical weeks: what’s happening with my body, is this normal, and what comes next.
The Problem
Early attrition
The dropout numbers were stark. Approximately 45% of enrolled patients left during the first week of the program. Of those who remained, roughly another 45% dropped out in week two. By the end of week three, roughly a quarter of the remaining cohort had also disengaged. The program was losing the majority of its participants before they reached the content designed to help them.
Root cause analysis
Two primary drivers emerged through coaching observations, qualitative feedback from the quality assurance team’s patient interviews, and my review of the clinical literature.
Dose escalation confusion. Many patients had no idea that dose escalation was part of their treatment plan. Their prescribers hadn’t explained the process, so patients believed the starter dose was the permanent dose. They didn’t know that dosage would increase over time, that different doses might require different delivery formats, or that the escalation process was expected and clinically intentional. When changes happened, patients interpreted them as errors or cause for concern rather than as normal progression.
Side effect anxiety. Gastrointestinal side effects are common in the early weeks of GLP-1 therapy and during dose escalation. But without context, patients experiencing nausea, reduced appetite, or digestive discomfort interpreted these symptoms as signs that something was wrong. Many discontinued the medication on their own, without consulting their care team, because they didn’t know the effects were typically temporary and would diminish as their bodies adjusted.
Both drivers pointed to the same structural gap: patients were starting a new medication without the foundational knowledge they needed to stay the course, and the program’s unbranded format meant it couldn’t fill that gap directly.
Constraints and Regulatory Environment
Any intervention had to operate within strict pharmaceutical content regulations. The team could not make direct medical recommendations or advise patients to change their medication behavior. Adverse events had to be flagged and routed appropriately; side effects warranting medical attention could not be minimized or omitted. All content required Medical-Legal-Regulatory (MLR) review and approval before deployment.
Beyond standard compliance, this project introduced a novel regulatory challenge: branded content had never been embedded within a white label program structure in this way. There was no established precedent, no existing template for what the review process should look like, and no shared understanding between the team and the MLR board about where the boundaries fell.
Fair balance requirements and promotional content rules directly governed what could and could not be said. This limited the scope of new content but also provided a useful constraint. The pharmaceutical client’s own patient-facing website content had already cleared regulatory review, which gave the team a roadmap: content that aligned closely with what the client had already published was safe territory, while anything that ventured beyond it would draw scrutiny.
MLR aggressively flagged any language that could read as medical advice, even when supported by existing clinical literature. I learned quickly that the safest and most productive approach was to reformat and restructure tips, information, and guidance that the pharmaceutical company already provided on its consumer-facing site, adapting it for the coaching program’s format and delivery model rather than writing entirely original clinical content.
The Intervention
Design approach
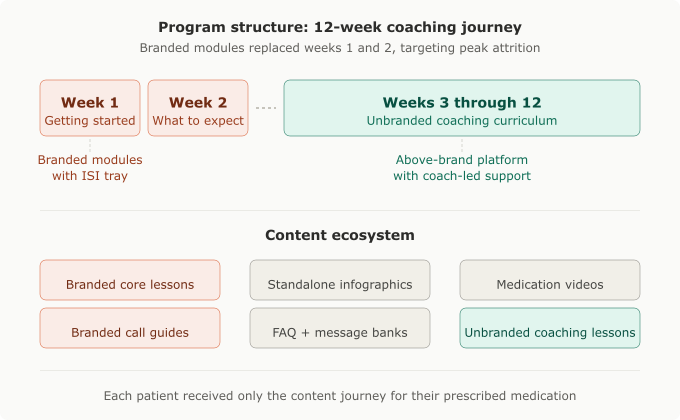
I designed two branded educational modules to replace existing content at the beginning of the twelve-week program, targeting the exact window where attrition was highest.
Week 1: Getting Started. This module replaced the program’s original onboarding lesson. It introduced the coaching program in the context of the patient’s specific medication, covering onboarding information for their GLP-1 therapy, the importance of dose escalation and adherence, and an initial assessment of where the patient was in their diabetes journey. It also incorporated branded content on managing common side effects, giving patients a framework for understanding early physical responses to the medication. The goal was to address dose escalation confusion and side effect anxiety before either became a reason to leave.
Week 2: What to Expect and Overcoming Barriers. This module replaced the original goal-setting lesson. It educated patients on what to expect as they continued GLP-1 therapy, addressed common barriers to medication adherence, and provided information about dose escalation and other possible dose changes. Content on overcoming medication-specific barriers, originally scoped for later in the program, was consolidated here to reach patients during the critical second-week dropout window. The goal was to normalize the early experience and give patients a framework for understanding what was happening, reducing the anxiety that was driving week-two attrition. Goal-setting content from the original lesson was delivered separately as unbranded supplemental material via email or SMS.
From week three onward, the program continued with the original unbranded coaching curriculum. The decision to concentrate all branded content in the first two weeks was deliberate: it addressed the identified attrition drivers at the point of highest risk, then allowed patients to transition into the broader lifestyle and diabetes management content with a stronger foundation of medication-specific knowledge.
The broader content ecosystem also expanded to include updated coaching call guides with medication-specific information, branded FAQ updates, message bank updates for coach-to-patient communications, standalone branded infographics, and medication-specific videos.
Delivery and user experience
Every lesson URL was unique to the individual patient and delivered via email or text from their assigned health coach. Branded lessons in weeks one and two carried the medication’s branding and included a collapsible Important Safety Information (ISI) tray. From week three onward, the program transitioned to the client’s above-brand educational platform, maintaining the coach-led relationship but shifting away from medication-specific branding.
Parallel content journeys were built for each medication the client supported, and they were never presented together. A patient received only the content relevant to their prescribed treatment.
Coaching calls followed a similar model. The first branded call included a full ISI reading, with subsequent calls offering the ISI and reading it again at the patient’s request. This approach had been developed in collaboration with the client’s safety and regulatory team and had been in use for over a year in a related coaching program, providing the regulatory precedent needed for approval.
The core design challenge
The central tension in this project was communicating accurate, reassuring information about dose escalation and side effects without crossing into medical advice or minimizing adverse events that warranted escalation to a healthcare provider or adverse event reporting system.
Resolving this tension was not a single design decision but an ongoing negotiation across multiple MLR review sessions. As the point person for MLR review, I worked iteratively with the review board to define what best practices looked like in this novel context: how much information was too much, which scientific references were considered safe sources, where the guardrails sat, and what patients actually wanted to know versus what could be said within promotional content rules. The framing that emerged used universal patient language, health self-efficacy principles, and a consistent “what to expect” structure, giving patients a sense of agency and preparedness without prescribing specific medical actions.
Process and Timeline
The full process from initial scoping to launch took approximately sixteen months. The project moved through several distinct phases: a pre-discovery period following the client’s initial request; a discovery and literature review phase; content design; studio production of first-iteration drafts for MLR review, including articles, PDFs, and videos; submission of all assets to the Veeva Vault content library; multiple revision rounds; final production; technical integration; and launch.
The initial timeline did not account for the level of MLR scrutiny involved in mixing branded content with a white label program structure. This was uncharted territory for the review board, and early rounds surfaced fundamental questions about scope and format that had to be resolved before content-level feedback could even begin.
I had initially expected that careful, evidence-based additions to the program, grounded in clinical literature, would be well received by the review board. That assumption proved optimistic. While I had correctly anticipated that heavy branding and promotional medication claims would not survive MLR review, I underestimated how narrowly the board would define acceptable content in this novel format. The client initially pushed for more original medication-specific material, but my early instinct that over-reliance on branding and medication benefit claims would draw rejection proved accurate. Over the course of many months and multiple review cycles, the project converged on a scope closer to my original recommendation: branded content built primarily from the client’s existing approved resources, restructured and reformatted for the coaching program’s delivery model, with original content limited to what could pass the high bar the MLR board had set for this new format.
Outcome
MLR approval was obtained, and the branded modules launched within the white label coaching program, establishing a new precedent for this type of hybrid content structure in pharmaceutical digital health coaching.
Early signals were positive. Coaches reported more engaged early conversations, patients provided favorable feedback, and client representatives were extremely satisfied with the launched product. The client’s team was already discussing next steps, including updating the branded modules to reflect upcoming product changes. Continued participation numbers were improving. Post-launch quantitative data is not available due to organizational changes at both the coaching company and the client that occurred shortly after launch.
The primary value of this project lies in the process itself: identifying a structural gap in how patients were being supported, designing a novel intervention within tight regulatory constraints, and successfully navigating an approval process with no established precedent.
Lessons and Implications
Regulatory timelines for novel content formats are unpredictable, and that’s worth planning for. This project took sixteen months, not because the content was complex, but because the format was new. When branded content has never been embedded in a particular structure before, the MLR review process isn’t just reviewing content; it’s establishing the rules for what this type of content can look like. Teams proposing novel approaches should build in significant buffer time and treat early review rounds as scope-defining conversations rather than expecting pass/fail feedback on drafts.
Early regulatory consultation pays for itself. I flagged the likely scrutiny level early, and that assessment proved accurate. When the client’s initial request for heavier branding drew the expected pushback, the project had room to course-correct rather than starting over. Involving regulatory thinking at the strategy phase, not just the review phase, saves months.
Patient education is a medication adherence intervention. The root cause of attrition wasn’t dissatisfaction with the coaching program, which maintained a Net Promoter Score around 70%. It was a knowledge gap that existed before patients ever enrolled. Patients were discontinuing medication because no one had explained what to expect. Filling that gap with well-timed, well-structured educational content addressed a problem that no amount of coaching skill could solve on its own.
The tension between branded and white label content is a design problem, not just a compliance problem. The challenge wasn’t simply getting branded content approved. It was designing a user experience where branded modules and unbranded coaching content felt like a coherent journey rather than two programs awkwardly stitched together. The transition from branded lessons in weeks one and two to the client’s above-brand platform in week three had to feel natural from the patient’s perspective, even though it involved different content sources, different branding, and different regulatory requirements.
Prescribers aren’t consistently preparing patients for GLP-1 therapy, and that gap is creating downstream problems. The most striking finding from this project wasn’t about the coaching program at all. It was that patients were being prescribed a medication with a well-documented escalation process and well-documented early side effects, and they were arriving with no preparation for either. This is a systemic gap in how GLP-1 medications are introduced to patients, and it creates avoidable attrition for any program trying to support adherence downstream.